Quick Facts
- Category: Science & Space
- Published: 2026-05-04 05:23:25
- Uncovering the Hidden Productivity Killers in Your IT Ecosystem: A Step-by-Step Guide
- Exploring the GPD BOX: A Panther Lake Mini PC with Cutting-Edge PCIe 5.0 Connectivity
- 5 Critical Facts About the Takedown of Massive IoT Botnets
- 7 Things You Need to Know About the Python Security Response Team (PSRT)
- Nintendo Switch 2 Splatoon Raiders Preorder Price Slashed: Amazon and Walmart Shave 17% Off Physical Copies
Introduction: The Universal Genetic Code
The genetic code is one of the most fundamental and ancient features of life on Earth. With only minor variations, all known organisms use the same set of three-letter DNA sequences—codons—to specify the same 20 amino acids when building proteins. This remarkable consistency has led scientists to believe that the genetic code originated in the last universal common ancestor (LUCA) of all life. But how did this code evolve from simpler beginnings? Most hypotheses suggest that early life forms used far fewer than 20 amino acids, gradually expanding the repertoire over time. To test this idea, a team of researchers from Columbia University and Harvard University decided to see if they could eliminate one of the 20 amino acids from the modern code—specifically, isoleucine—and still have a functioning ribosome. Their work provides a fascinating glimpse into the flexibility of life’s basic machinery.
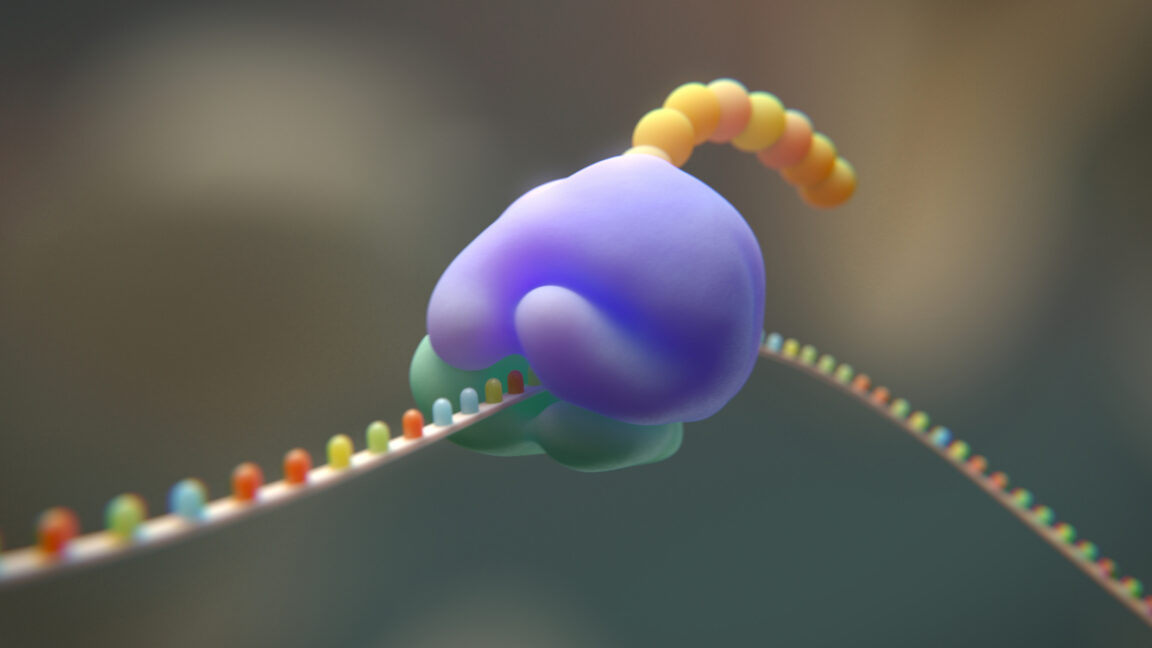
Background: The Puzzle of Code Evolution
The genetic code is not just a list of mappings; it is deeply integrated into the cellular machinery that translates DNA into proteins. The ribosome, a complex molecular machine, reads mRNA codons and links together amino acids in the correct order. Each amino acid is brought to the ribosome by a specific tRNA molecule, and the process requires precise interactions. The 20 amino acids we see today have diverse chemical properties—some are hydrophobic, others charged, and some small and flexible. How did such a system emerge from a simpler one?
One prevalent theory is that the earliest genetic codes used only a handful of amino acids, perhaps those that could be synthesized easily under prebiotic conditions, such as glycine, alanine, and aspartic acid. Over time, as metabolism became more complex, new amino acids were added, expanding the code. But direct evidence for these early stages is scarce. By attempting to reduce the code now, researchers can test whether such a reduction is even possible and gain insights into its original constraints.
The Experiment: Removing Isoleucine from the Ribosome
Why Target Isoleucine?
Isoleucine is an essential amino acid—humans cannot synthesize it—and it plays a critical role in protein structure, often found in hydrophobic cores. The team chose isoleucine for its first attempt because its removal would be a severe test. If a key component of the translation machinery could function without isoleucine, it would demonstrate a remarkable degree of built-in flexibility.
Engineering a Modified Ribosome
The researchers focused on a specific region of the ribosome that normally contains isoleucine. Using genetic engineering techniques, they replaced the relevant codons with sequences coding for other amino acids, effectively removing isoleucine from that ribosomal protein. Remarkably, the modified ribosome was still able to assemble and function, albeit with reduced efficiency. This indicates that the ribosome, and by extension the genetic code, may have some tolerance for amino acid substitutions.
This experiment is a proof of concept: if one essential amino acid can be removed from a critical component, perhaps the entire code can be slimmed down. However, the researchers caution that they only modified a single ribosomal protein, not the entire set of cellular proteins. Still, the result opens the door to further reductions.
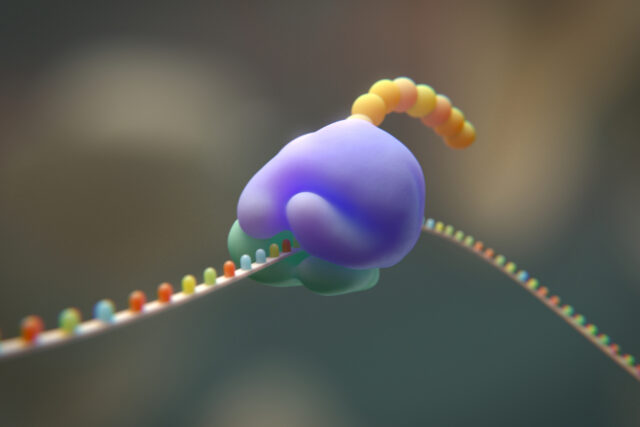
Implications and Future Directions
Testing Evolutionary Hypotheses
The immediate implication is that a 19-amino-acid code is plausible, supporting the idea that early life could have operated with fewer building blocks. The team plans to continue removing other amino acids, one by one, to see just how minimal a functional genetic code can be. If successful, these experiments would provide empirical evidence for stepwise evolution of the code.
Applications in Synthetic Biology
Beyond pure evolution, this work has practical applications. If researchers can streamline the genetic code, they might create organisms that use non-standard amino acids for novel functions—such as producing new materials or drugs. By freeing up codons that were previously used for natural amino acids, scientists can reassign them to biosynthetic pathways for synthetic molecules. This could revolutionize biotechnology, allowing for enzymes with enhanced stability or entirely new catalytic activities.
Challenges and Limitations
Removing an amino acid from a single ribosomal protein is one thing; removing it from every protein in an organism is another. Isoleucine appears in thousands of proteins, each potentially essential for life. The current experiment only shows that a specific ribosome component can adapt. The next step would be to engineer an entire bacterium that lacks isoleucine from its proteome—a monumental task that may require extensive rewiring of metabolic and regulatory networks.
Conclusion: A Step Toward Understanding Life's Origins
The Columbia-Harvard team’s work is a bold attempt to rewrite the fundamental rules of biology. By showing that the ribosome can function without isoleucine, they have taken the first step toward a 19-amino-acid genetic code. This not only supports theories of an early, simpler genetic system but also paves the way for synthetic organisms with expanded capabilities. As research progresses, we may eventually see a living cell with a reduced set of amino acids, offering a window into the deep past of life on Earth and a tool for engineering the future.